Why This Matters Now
When a laundry pod dissolves in cold water, the first thing that must happen is wetting. The surfactant needs to get the water to penetrate the fabric and reach the soil. If the surfactant can't lower surface tension fast enough at 15°C, the cleaning suffers. No amount of enzyme optimization or fragrance work will fix it.
This is not a new problem. Surface tension has always mattered; what's changed is the margin for error.
Ten years ago, most laundry detergents used similar surfactant systems with decades of optimization behind them. Today, brands are reformulating with bio-based surfactants, cutting concentrations for sustainability, and trying to make products work in cold water. Each of those changes affects the surface tension curve in ways that aren't always obvious until the product fails in the field.
The same story is playing out across industries. Personal care is moving away from sulfates, agrochemical companies are reformulating for drone applications at ultra-low volumes, pharmaceutical manufacturers are trying to keep proteins stable through fill-finish operations, and coatings formulators are switching from solvent-based to water-based systems. In every case, surface tension sits at the center of the problem.
The surfactants market is worth over $45 billion globally and continues to grow. But that growth is coming with constraints that make interfacial design harder, not easier.
Trends Driving Reformulation

Several pressures are hitting multiple industries at once, and they all affect how formulators think about surface tension:
Bio-based and sustainable surfactants. Whether it's sophorolipids in home care, alkyl polyglucosides in personal care, or natural emulsifiers in food, the shift away from petroleum-derived ingredients is accelerating. These alternatives have their own surface tension profiles that don't match conventional ingredients. A formulation optimized for sodium laureth sulfate won't work the same way with sodium cocoyl isethionate. A dressing stabilized with polysorbate 60 won't behave the same with sunflower lecithin.
Concentration and reduced water. Concentrated products save packaging, transportation, and storage costs. They also reformulate the problem. A product designed to work at 10% surfactant operates in different territory on the surface tension curve than one designed for 2%. Concentrated laundry pods, solid shampoo bars, and waterless cleaning products all require rethinking how surface tension develops during use as the product dilutes or activates.
Cold and low-energy processing. Cold-water laundry, low-temperature cleaning, ambient-temperature coating processes. Reducing energy use often means working at temperatures where surfactants are less soluble and adsorb more slowly. Formulations optimized for warm conditions may not translate.
Precision application. Drone spraying delivers pesticides at 1-5 liters per hectare instead of 100-400 liters for ground spraying, so every droplet has to work. High-speed inkjet printing creates droplets in microseconds, and targeted drug delivery puts tight tolerances on droplet size and behavior, requiring exact interfacial control.
Mildness and biocompatibility. Consumers want gentler products. Regulations require better safety profiles. The surfactants that meet these requirements often have larger molecular structures with slower adsorption kinetics. Achieving the same functional performance with milder ingredients requires reformulation, not just substitution.
Regulatory restrictions. PFAS bans affect coatings, textiles, and food packaging, while ongoing reviews of surfactant safety affect multiple categories. When an ingredient gets restricted, the replacement rarely has identical surface tension behavior and reformulation is required.
These trends explain why surface tension has moved from a background property to a front-and-center formulation challenge. Understanding the underlying science is the first step to addressing it.
Surface Tension Basics
The Physics
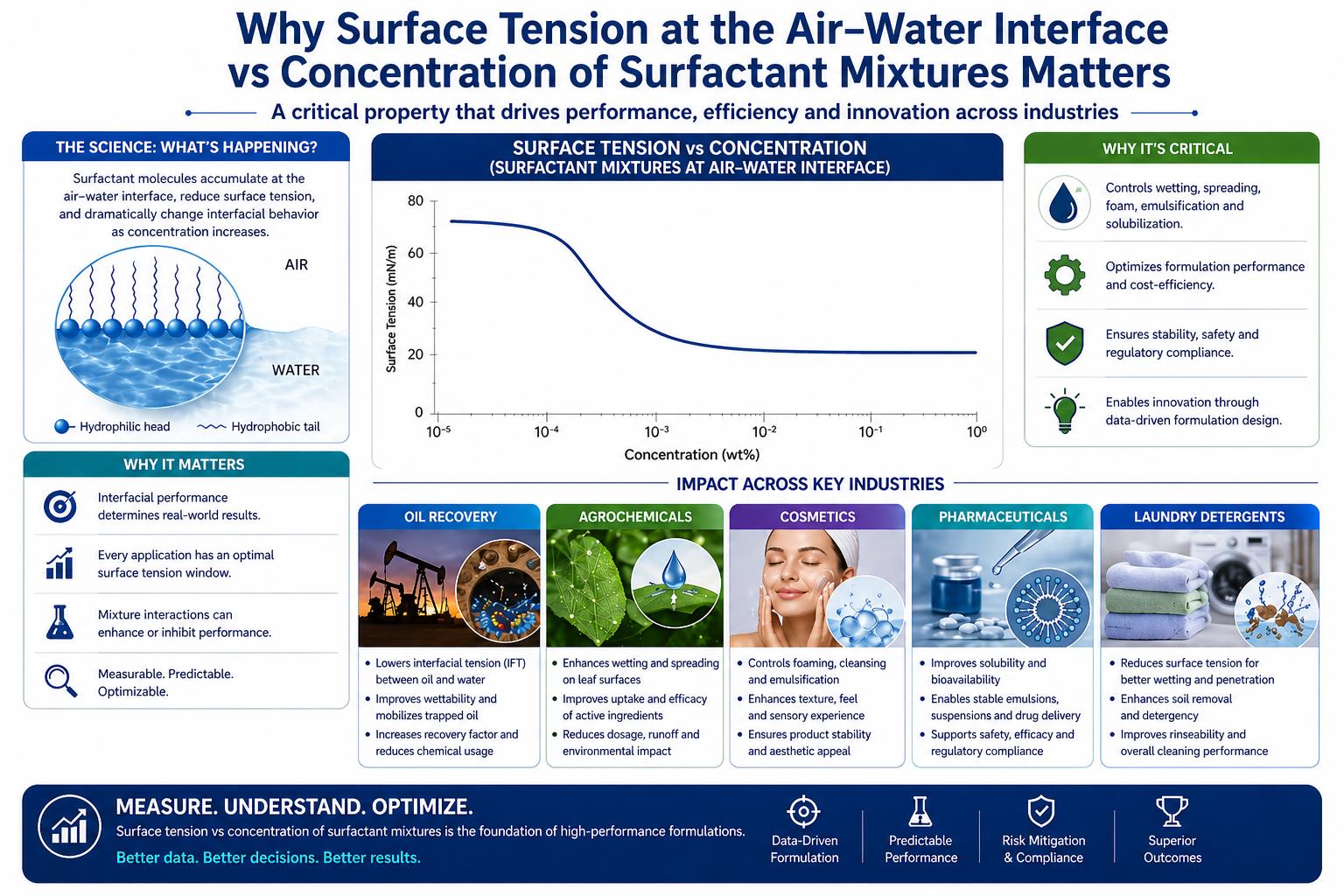
Water molecules attract each other through hydrogen bonding. In the bulk of the liquid, a water molecule feels these attractions from all sides. At the surface, there's nothing above it. The molecule gets pulled inward and sideways, but not up. This imbalance creates tension at the surface, like a stretched membrane.
Pure water at 25°C has a surface tension around 72 mN/m. That's high compared to most organic liquids. It's why water beads up on a waxed car instead of spreading, and why water droplets are spherical.
What Surfactants Do
Surfactants are molecules with a water-loving head and a water-hating tail. Drop them in water and they migrate to the surface, tails pointing up into the air, heads staying in the water. This disrupts the hydrogen bonding network at the interface and lowers surface tension.

The relationship between surfactant concentration and surface tension follows a predictable pattern: At low concentrations, not enough surfactant reaches the surface to matter much. As concentration increases, more surfactant packs into the interface and surface tension drops. This continues until you hit the Critical Micelle Concentration (CMC). At that point, the surface is saturated. Add more surfactant and it forms micelles in the bulk instead of going to the surface. Surface tension plateaus.
The CMC matters because below it, surface tension is sensitive to small concentration changes. Above it, surface tension is stable but you're paying for surfactant that isn't doing interfacial work.
Mixed Systems
Nobody uses single surfactants in real products. A typical formulation might have an anionic primary surfactant, a nonionic co-surfactant for mildness, and an amphoteric for foam boosting, which don't behave independently.
Mixed surfactant systems often show synergy. The blend achieves lower surface tension than either component alone at the same total concentration. The different molecular shapes pack together more efficiently at the interface.
The problem is that this synergy is hard to predict since the composition at the surface isn't the same as the composition in the bulk. Change one surfactant in a blend, and the whole surface tension profile can shift in unexpected ways.
Equilibrium vs. Dynamic
Lab measurements usually report equilibrium surface tension. You put your sample in a tensiometer, wait for the reading to stabilize, and record the number. This can take seconds to minutes depending on the surfactant.
Real applications don't always wait for equilibrium. For example, when a spray nozzle atomizes a pesticide into droplets, the process happens in milliseconds. The surfactant doesn't have time to fully populate the fresh droplet surface. Thus, dynamic surface tension (what you get before equilibrium) is higher, sometimes much higher.
A formulation can look great in equilibrium measurements and fail in the field because the surfactants are too slow to adsorb during actual use. This becomes especially relevant when switching to milder or bio-based surfactants, which tend to have bulkier molecular structures and slower kinetics.
Why Surface Tension Matters for Products
Surface tension affects formulated products at three stages: during manufacturing, at the moment of application, and over the product's lifetime. The specific effects depend on the product type, but the underlying physics is the same.
Wetting and Spreading
A liquid can only do its job if it contacts its target surface. Water's high surface tension works against this. Without surfactants, water beads on skin, fabric, leaves, metal, plastic, and most other surfaces instead of spreading into a thin film.
The target surface tension depends on the substrate. Spreading occurs when the liquid's surface tension is lower than the substrate's surface energy. Waxy plant leaves have surface energies around 20-30 mN/m, and getting water-based sprays to spread on them requires aggressive surface tension reduction. Human skin has higher surface energy, making wetting easier but still requiring some surfactant for good spreadability.
Droplet Formation and Spray Behavior
When liquids pass through nozzles or atomizers, surface tension determines droplet size. Higher surface tension resists the breakup of the liquid stream, producing larger droplets. Lower surface tension allows finer atomization.
For agricultural spraying, this is a balancing act. Smaller droplets give better coverage but drift more in wind. Larger droplets are less prone to drift but may not cover the target adequately. The optimal droplet size distribution depends on the crop, the pest, and the application conditions. Surface tension is one of the main levers for controlling it.
The same physics applies to spray coatings, inkjet printing, and any other process that forms droplets. In pharmaceutical fill-finish, droplet formation during filling can create foam or introduce air that causes protein aggregation. Controlling surface tension helps manage these effects.
Foam Generation and Stability
Foam is a dispersion of gas in liquid, stabilized by surfactant films at the bubble surfaces. Whether foam is desirable depends on the product. Shampoos and dish soaps are expected to foam, while laundry detergents for front-load washers shouldn't foam too much. Industrial processes often need defoaming.
Surface tension affects foam generation: lower surface tension makes it easier to create the large interfacial area that foam represents. However, foam stability depends more on the properties of the interfacial film: its thickness, its elasticity, and how well it resists drainage.
Reformulating with different surfactants changes both the generation and stability of foam. A sulfate-free shampoo might generate foam more slowly but have foam that persists differently during rinsing. Getting the foam profile right requires understanding how the new surfactant system behaves at air-water interfaces.
Emulsion Formation and Stability
Emulsions are dispersions of one liquid in another, stabilized by surfactants at the droplet surfaces. The interfacial tension between oil and water determines how much energy is needed to create small droplets during homogenization and how thermodynamically unfavorable the emulsion is.
Lower interfacial tension makes emulsion formation easier and can improve stability by reducing the driving force for coalescence. But stability also depends on the mechanical properties of the interfacial film and any electrostatic or steric barriers between droplets.
This matters for personal care products like lotions and creams, for food products like dressings and sauces, for pharmaceutical emulsions, and for many industrial applications. The shift toward natural emulsifiers in food and personal care has made emulsion work harder because these ingredients form different interfacial films than synthetic alternatives.
Protein Stability at Interfaces
Proteins are surface active, so they adsorb to air-water and oil-water interfaces, where they can unfold and aggregate. This is a problem for biologic drugs, where the therapeutic protein needs to stay in its native conformation.
During manufacturing, filling, and shipping, protein solutions encounter mechanical stresses that create fresh interfaces. Pumping, filtration, filling, and even agitation during transport can cause interfacial damage. Surfactants are added to compete with the protein for interfacial sites, protecting the therapeutic molecule.
This protective function depends on the surfactant getting to the interface faster than the protein. Dynamic surface tension behavior matters more than equilibrium values. The surfactant concentration needs to be high enough to provide protection but not so high that it causes other stability issues.
The Coupling Problem
Surface tension doesn't exist in isolation. Change your surfactant system and you change:
- Viscosity (surfactants form micelle networks, interact with polymers)
- Stability (surfactants stabilize emulsions and dispersions)
- Phase behavior (surfactant concentration determines micelle shape and liquid crystal formation)
- Foam (surface tension affects generation, interfacial rheology affects stability)
A formulation that hits target surface tension but fails on viscosity isn't a solution, and neither is one that has great surface tension but poor stability. All properties must be in the targeted ranges for the product to function properly.
The variables that affect surface tension include:
- Surfactant type and concentration
- Co-surfactant ratios
- Salt content
- pH
- Temperature
- Polymer additives
- Oil phase composition
Traditional DOE can map some of this space, but it scales poorly. Three surfactants at five levels each with two co-additives at three levels gives you 375 combinations before you even consider temperature or pH. Real formulation spaces are much larger.
Where Predictive Tools Fit
The point of predictive formulation isn't to replace lab work. It's to make lab work more efficient by narrowing down where to look.
For surface tension, this means predicting how the surface tension vs. concentration curve will change when you substitute one surfactant for another, adjust a ratio, or add a co-additive. If you can screen 500 combinations computationally and identify the 20 most likely to work, you've saved months of experimentation.
To obtain high-quality predictions, chemistry-aware models trained on actual physicochemical data are needed. Models that learn the relationships between molecular structure, concentration, and interfacial behavior from experimental measurements.
FastFormulator takes this approach with a Virtual Surface Tensiometer that predicts surface tension vs. concentration curves for surfactant mixtures. Combined with the Virtual Viscometer and Virtual Stability Chamber, it allows exploration of formulation space before committing to synthesis and testing.
The companies that do well in the current environment won't be the ones running the most experiments, they'll be the ones running the right experiments.

Takeaways
Surface tension determines whether products wet, spread, foam, emulsify, and atomize correctly. As industries reformulate for sustainability, mildness, and concentration, the comfortable surfactant systems of the past are being replaced with alternatives that have different interfacial behavior. Getting surface tension right has become harder, not easier.
The difficulty is that surface tension is tangled up with everything else in a formulation. You can't optimize it independently of viscosity, stability, and phase behavior. The variables interact in ways that make exhaustive experimental mapping impractical within real development timelines.
Predictive tools built on physicochemical modeling can help by identifying promising formulation candidates before experimental work begins. This isn't about removing human judgment from formulation. It's about pointing that judgment toward the regions of formulation space most likely to contain solutions.
References
- Rosen, M.J. and Kunjappu, J.T. (2012). Surfactants and Interfacial Phenomena, 4th Edition. John Wiley & Sons, Hoboken, NJ.
- Eastoe, J. and Dalton, J.S. (2000). Dynamic surface tension and adsorption mechanisms of surfactants at the air-water interface. Advances in Colloid and Interface Science, 85(2-3), 103-144.
- Holland, P.M. and Rubingh, D.N. (1992). Mixed Surfactant Systems. ACS Symposium Series 501, American Chemical Society, Washington, DC.
- Chang, C.H. and Franses, E.I. (1995). Adsorption dynamics of surfactants at the air/water interface: a critical review of mathematical models, data, and mechanisms. Colloids and Surfaces A, 100, 1-45.
- Grand View Research (2023). Surfactants Market Size, Share & Trends Analysis Report, 2023-2030.
- Van Bogaert, I.N.A., et al. (2007). Microbial production and application of sophorolipids. Applied Microbiology and Biotechnology, 76(1), 23-34.
- Ozturk, B. and McClements, D.J. (2016). Progress in natural emulsifiers for utilization in food emulsions. Current Opinion in Food Science, 7, 1-6.
- Carpenter, J.F., et al. (2009). Overlooking subvisible particles in therapeutic protein products: gaps that may compromise product quality. Journal of Pharmaceutical Sciences, 98(4), 1201-1205.
- Hilz, E. and Vermeer, A.W.P. (2013). Spray drift review: the extent to which a formulation can contribute to spray drift reduction. Crop Protection, 44, 75-83.
- Wicks, Z.W., et al. (2007). Organic Coatings: Science and Technology, 3rd Edition. John Wiley & Sons, Hoboken, NJ.