
Stability Used to Be Simple
For decades, stability testing followed a predictable workflow. A formulation was prepared, distributed into containers, and placed into temperature-controlled environments at various temperatures based on the industry standard, examples could be like 5°C, 25°C, and 50°C. Samples were observed over time for signs of separation, aggregation, viscosity drift, or visual degradation. If a formulation failed, the process restarted with a modified composition. This approach is still widely used today. Even under "accelerated" conditions at 40°C, meaningful results typically can take at least a month, while real-time testing at ambient conditions can stretch to four to six months.
Colloidal stability directly governs whether a product performs as intended over time. Phase separation, sedimentation, or aggregation do not just affect appearance, they change dosing, application behavior, and functional efficacy. A coating that separates will not apply uniformly. An agrochemical that sediments will deliver inconsistent active concentration. A cosmetic that drifts in texture will be rejected by consumers. Stability is not separate from performance. It is the condition required for performance to persist from production to end use.
This workflow assumes that time is available. Increasingly, it is not. The question is not whether stability testing works, but whether it fits within the timelines that modern formulation teams are expected to operate under.
What Is Actually Changing?

Across formulation-driven industries, multiple structural shifts are happening at once. Products are becoming more complex, incorporating a greater number of functional ingredients with interacting roles. At the same time, there is strong pressure toward sustainability, which often means replacing well-characterized synthetic components with bio-based or naturally derived alternatives. On top of that, performance expectations continue to rise, with products expected to deliver precise behavior under increasingly demanding conditions. Each of these trends independently makes stability more difficult. Together, they create systems that are fundamentally harder to control.
These pressures show up differently depending on the application, but they lead to the same outcome. In cosmetics and consumer products, clean-label formulations replace robust synthetic stabilizers with natural ingredients that introduce variability and weaker interfacial behavior, while higher sensory expectations make even minor instability unacceptable. In agrochemicals, higher active concentrations and precision application methods such as drone spraying increase the risk of sedimentation, aggregation, and nozzle clogging, with tank-mix compatibility adding further complexity. In coatings, the shift to waterborne systems for VOC compliance makes dispersions more prone to flocculation and phase separation, directly impacting finish quality. In pharmaceuticals and biopharma, proteins, biologics, and advanced delivery systems such as nanoparticles introduce instability pathways with direct safety and regulatory consequences. In food and beverage systems, plant-based ingredients and clean-label constraints remove effective stabilizers while maintaining expectations for consistent texture and appearance.
The common thread is not any one industry shift, but the convergence of all of them. Formulations are being pushed toward higher complexity, tighter constraints, and more demanding performance standards, all while becoming inherently harder to stabilize. Stability is no longer a secondary concern. It is the limiting factor that determines whether these innovations can survive in real-world conditions.
Why Formulations Become Unstable
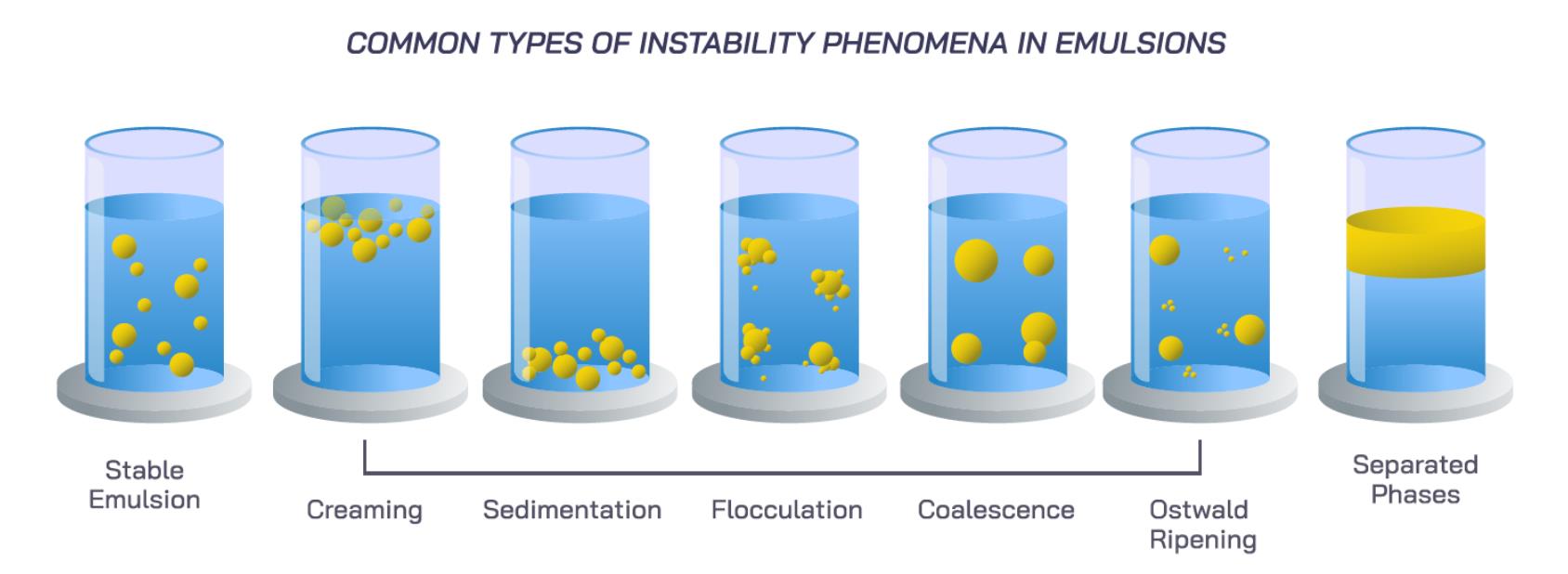
At a fundamental level, most colloidal systems are not stable in the thermodynamic sense. Emulsions, suspensions, and dispersions exist in a metastable state. They are created through energy input that disperses one phase into another, forming droplets or particles. The natural tendency of the system is to minimize interfacial area and return to a separated state. Stability is therefore not about achieving a permanent equilibrium. It is about slowing down the pathways that lead to separation.
These pathways are well understood. Sedimentation or creaming occurs when there is a density difference between the dispersed and continuous phases. Under gravity, droplets either settle or rise, depending on their relative density. The rate of this process depends on particle size, density difference, and the viscosity of the surrounding medium. Larger droplets and lower viscosity accelerate separation.
Flocculation is often an early-stage instability where droplets cluster together without fully merging. This occurs when attractive forces between particles overcome repulsive barriers. While flocculation can sometimes be reversible, it increases the likelihood of further instability. Coalescence follows when droplets merge into larger ones, reducing total interfacial area. Unlike flocculation, coalescence is irreversible and directly drives phase separation.
Ostwald ripening introduces another mechanism, particularly in fine emulsions. Smaller droplets dissolve into the continuous phase and redeposit onto larger droplets due to differences in internal pressure. Over time, this leads to a coarsening of the droplet size distribution, which accelerates other forms of instability. In extreme cases, systems can undergo phase inversion, where the dispersed and continuous phases effectively swap roles.
Underlying all of these behaviors is a balance of opposing forces. On one side, particles naturally attract each other due to weak universal forces that are always present. On the other side, stability comes from forces that keep particles apart and prevent them from sticking together. These repelling effects can come from electrical charge on the particle surface or from layers of molecules attached to the surface that act like a physical barrier.
When repulsion is strong enough, particles stay separated and the system remains stable. When it is weak, particles come close, stick together, and eventually form larger clusters that can lead to separation.
Scientists often describe this balance using a framework called DLVO theory, and in practice they use measurements like zeta potential as a rough indicator of how strong the electrical repulsion is. Higher values generally mean stronger repulsion and better resistance to aggregation.
Instability is not random. It follows from these underlying physical principles. The challenge is that in complex, multi-component formulations, these forces are influenced by a wide range of interacting variables.
Engineering Stability
Because instability comes from identifiable mechanisms, formulators can address it by directly changing ingredients and processing conditions. Here are some of the routes that can be taken:
Oil-Water Interface Engineering
The most direct lever is the droplet interface. Formulators add emulsifiers, which are ingredients that sit at the boundary between oil and water, reduce interfacial tension and help keep droplets from merging. They work by coating droplets and reducing the tendency for them to stick together. Their effectiveness depends on how completely they cover the surface. If coverage is uneven, droplets can still find exposed areas and aggregate. In more robust systems, formulators may choose different emulsifiers, combine multiple surfactants, or use polymer or particle based stabilizers that form a stronger layer around droplets. Selection often depends on matching the emulsifier system to the oil water balance of the formulation and the other ingredients present as captured by HLD.
Electrostatic Stabilization
Another key lever is electrostatic stabilization, which is controlled through ingredient choice and conditions such as pH. Changing pH can alter whether certain components carry charge, which directly affects how strongly droplets repel each other. Adding salts such as NaCl reduces this effect because ions in solution shield surface charges and make it easier for droplets to come into contact. Steric stabilization works differently. It relies on adding polymer ingredients that extend into the surrounding liquid and create a physical barrier that prevents close approach. This approach is often less sensitive to pH and salt, but it depends on selecting polymers that adsorb strongly to the droplet surface and remain attached over time.
Viscosity Modification
Formulators also adjust stability through the properties of the continuous phase. Adding thickeners increases viscosity, which slows droplet movement, reduces collisions, and slows separation. In some systems, structure forming ingredients are used to create a yield stress so droplets remain effectively fixed unless the product is strongly disturbed.
Droplet Size
Droplet size is another major control point. It is set through processing conditions such as mixing intensity, shear, and homogenization pressure. Smaller droplets are generally more stable because they are less likely to merge and separate under gravity, which is why high shear processing is widely used.
Each of these changes comes with trade offs. Increasing viscosity can change how a product feels or spreads. Adjusting pH to improve stability can affect sensitive active ingredients. Increasing high shear to reduce droplet size can increase processing costs. Improvements in stability are rarely isolated and often shift other parts of the system.
This creates a central tension. Formulations are constantly adjusted to meet performance and regulatory requirements, but each adjustment changes stability and requires revalidation.
The Speed Problem
The difficulty is not only in achieving stability but in confirming it. Traditional stability testing operates on timelines that are increasingly incompatible with modern product development cycles. Real-time testing can require several months before meaningful conclusions can be drawn. Accelerated testing reduces this to weeks, but still requires sequential evaluation of candidate formulations. A failed result means returning to the formulation stage and repeating the process.
This workflow is inherently reactive. A formulation is created, then tested, and only after a delay does its viability become clear. There is no built-in mechanism to assess, before experimentation, which formulation is most likely to succeed. When the design space is large, testing every possibility is impractical. Teams are forced to rely on intuition to narrow down candidates, but intuition struggles in systems where interactions are complex and non-linear.
The business implications are direct. In many industries, reformulation cycles that once spanned over a year now need to be completed in a matter of months. Stability testing alone can consume a significant fraction of that timeline. As product complexity increases and timelines compress, the gap between what is required and what traditional workflows can deliver continues to widen.
A Better Way to Know Before You Make

This is exactly the problem FastFormulator was built to address.
Rather than treating stability as a downstream property that is measured after a formulation is created, we approach it as a function of formulation composition and underlying chemistry. The mechanisms that drive instability are well understood at a physical level. Sedimentation, flocculation, and coalescence are not arbitrary outcomes. They emerge from interactions between ingredients, structures, and conditions that can, in principle, be learned.
To capture this, we built chemistry-aware machine learning models trained on a large, proprietary dataset of real formulations. These models are designed specifically to learn relationships between chemical structure, formulation composition, and physical outcomes, including stability behavior across complex, multi-component systems. The goal is not to replace experimental validation, but to make it far more targeted and efficient.
In practice, this changes how formulation teams work. Instead of preparing ten candidate formulations and committing all of them to a month of accelerated stability testing, formulators can evaluate those candidates upfront. The system can identify which formulations are likely to remain stable and which are likely to fail through mechanisms like phase separation or aggregation. Experimental work can then be focused on the most promising candidates, reducing both the number of iterations and the time required to converge.
This approach has been validated across a range of full product formulations, where predicted stability outcomes align with observed experimental behavior. Critically, the system is not static. In addition to being trained on a large base of multi-component formulation data, the models can be further refined using a company's own experimental results. Historical stability studies, internal formulation databases, and even small batches of new experimental data can be incorporated to tune the model toward the specific chemistries, raw materials, and processing conditions that a given team works with.



Stability also does not exist in isolation. It is tightly coupled to other properties such as rheology. Changes that impact continuous phase viscosity, for example, directly influence sedimentation rates and droplet interactions. Because FastFormulator is built on a unified understanding of formulation chemistry, it can provide insight across these properties simultaneously, giving a more complete view of how a formulation will perform over time.
Conclusion
Stability is no longer a final checkpoint at the end of development. It is a central design variable that must be considered from the outset. The forces driving formulation complexity are not slowing down, and the demands placed on stability are increasing across every major industry.
The ability to anticipate stability behavior before running an experiment represents a shift from reactive testing to informed design. As with rheology, understanding and predicting stability enables more efficient exploration of formulation space. In modern formulation science, stability is not just about shelf life. It is the bottleneck between innovation and market success.
References
- Israelachvili, J.N. Intermolecular and Surface Forces. Academic Press, 3rd ed., 2011.
- Tadros, T.F. Emulsion Science and Technology. Wiley-VCH, 2009.
- Cox, R. Emulsions, SCS Summer School Lecture, 2014.
- McClements, D.J. Food Emulsions: Principles, Practices, and Techniques. CRC Press, 3rd ed., 2015.
- Malvern Panalytical. Zeta Potential: An Introduction in 30 Minutes.
- Soft Matter, 2014, 10, 6941–6954.